2. Material
2.1. Study sites
Investigations of climate and arthropod colonisation
Research was conducted from August 1993 until April 1994 in five areas near Kiel (northern Germany, hilly landscape, oceanic-temperate climate, less than 50 m asl; average yearly precipitation: 650-800mm; average monthly temperatures: 0-16°C, RAABE 1987, DEUTSCHER WETTERDIENST 1967). Additionally, in August and September 1993, an area near Rostock was considered (flat and climatically more continental, north-east Germany). Trunks of large solitary trees (> 2.5 m circumference at a height of 1.5 m; at most approx. 40 % of the trunk could be shaded by crowns of other trees or by shrubs except during dawn or dusk) were investigated at heights between 0.8 and 1.8 m. Specimens of Quercus robur L. (oak), Fagus sylvatica L. (beech), Fraxinus excelsior L. (ash) and, except in one area, Tilia sp. (mainly platyphyllos SCOP.) (lime) were considered. I additionally differentiated oaks and beeches with a strong algal cover (> 70% of the bark surface). Moreover, extremely solitary oaks were considered, defined as either neighbouring to only one or no trees within a radius of 30 m or as planted in a scattered row on a very exposed hill, only covered by fields. Within each study area, trees were selected in order to minimise distances between the different tree types. Generally, solitary trees are a common feature in these rural areas of investigation, mainly along paths and avenues and in meadows. Solitary beech trees grow mostly in small park-like forests or in clearings.
The considered, most frequent microhabitat types were (Fig 1): 1. the fruticose (shrub-like) lichen species Evernia prunastri L., 2.-4. the crust-like lichens Lepraria incana L., Lecanora expallens ACH., Pertusaria albescens Choisy & Werner (rarely combined with other Pertusaria species), 5. algal crusts (Pleurococcus sp.), 6. very sparse algae/hyphae covers, only recognisable with a lens ("no cover"), 7. horizontal crevices (perpendicular to bark scales and always starting at the bark surface inwards; apart from this horizontal axis, these crevices can take any direction), 8. vertical crevices (situated between bark scales; not realised in beeches). In addition, mosses and foliose lichens were found, but they never covered more than 5% of the bark surface - even when only a narrow zone of equal momentary exposures (see 2.2.) was considered. Only on ash trees did the lichens Buellia punctata Massal. and B. grisovirens Almb. sometimes cover up to 20% of the respective zone of exposure.
Figure 1: Structure of the considered, frequent microhabitat types for optical sampling. The scales are indicated at the right. Horizontal crevices are given in three-dimensional view. Thalli of Evernia prunastri can vary strongly in size. The phycobiots and free algae are indicated as , the medulla mycobiots and free hyphae as , the dense cortex mycobiots as . In Evernia prunastri only a single, basal branch is drawn in cross section. Signatures are more dense in the more compacted, stiff thalli. The phycobiot layers are interwoven with mycobiot hyphae to a different degree.
Investigations on the morphogenesis of the lichen Evernia prunastri
Investigations were conducted at 24 sites, situated in the described landscape near Kiel. Evernia prunastri was growing on trunks of solitary trees (along avenues, in hedgerows, in clearings, at forest-edges) and was mainly investigated on oaks (Quercus robur), limes (Tilia spp) and sometimes beeches (Fagus sylvatica). Thalli were shrub-like, mostly standing out from the trunk up to 25 mm, branches were 0.5-4.0 mm wide and 0.2-0.5 mm thick. Thalli covered basal surfaces of 35-500 mm2 each.
2.2. Climate measurements and sampling of arthropods
From August 1993 to January 1994 45-50 trees were sampled per month (in 1993 30 % of them at night), in February and March 1994 28 trees each and in April 8 trees. Daytime excursions started at 8 a.m. or, in winter, from daybreak and lasted until at least two hours before sunset. Night excursions began at dusk and lasted for 6 to 9 hours. Excursions were shorter only when temperatures remained below -5°C for several days. This was rare. On such days most trunks were not populated by arthropods at all. Then, most animals were probably not able to react to momentary climates on trunks because (i) the animals' mobility was reduced or (ii) they were diapausing or (iii) the frozen soil surface and litter layer interrupted the immigration of animals onto trunks. Therefore, such trunks were not considered for the analyses of animal distribution (except for the description of seasonal abundance dynamics). Also the investigation of the momentary climatic patterns was pointless then, since the climatological study was focused on trunks as a habitat for arthropods.
Different study areas and tree species were sampled in a random order. Nine trees were sampled twice, but only after a period of at least three months.
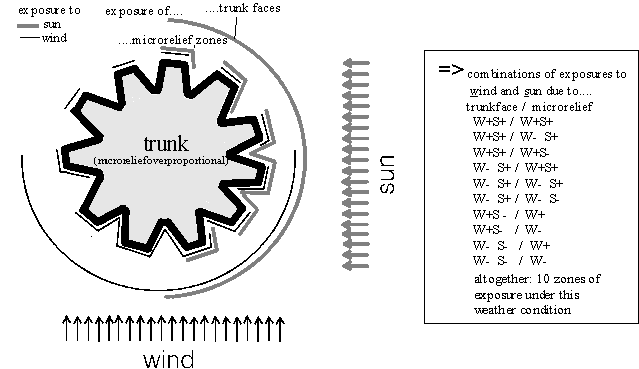
Each trunk was divided, by eye, into faces according to their exposure to momentary wind, sun and precipitation (= zones covered with a waterfilm). For instance, during windy, sunny weather (Fig. 2), normally four trunk faces were differentiated: those exposed to wind and sun, only to wind, only to sun or to none of them. Each trunk face was subdivided again into two to three zones according to different momentary exposures within its microrelief (the positions on either bark-ridges or on slopes of bark-valleys; shrub-like thalli could additionally shade parts of the nearby bark). "Zones of exposure" were defined according to the combination of exposures of both, the trunk face and the microrelief zone. In the example in Fig. 2, 10 zones of exposure were differentiated, whereas e. g. during windy, non-sunny, non-rainy weather there were only four zones.
In each zone of exposure the dominance of each of the considered epiphyte species was estimated according to their cover: <20% / >20 - <50% / >50 %.

Figure 2: Exposures of trunk faces and microrelief zones during windy, sunny weather (scheme, microrelief strongly enlarged and simplified). On the shaded trunk faces all incoming light is indirect, i. e. reflected or scattered light and thus reaches all parts of the microrelief in similar intensity. Therefore, no sun exposures could be differentiated within the microrelief of sun shaded trunk faces. In contrast, on wind-sheltered trunk faces there is still a clear decrease of air velocity from the bark ridges towards the bark valleys.
Climate measurements: Within each zone of exposure I measured temperature (Gultan Tastotherm D 700), air-velocity (thermic anemometer 641 N, Lambrecht Meßgeräte) and air-humidity (di-electrical sensors: Valvo-hygrometer, sensor with detached cap and detached ventilator; after February 1994: Rotronic hygrometer A1, with same precision and method, identical results, but faster response). The Valvo-sensor was mostly too large to measure along slopes of "valleys" in the microrelief, whereas the Rotronic sensor was sufficiently small in most cases. All measurements were taken at a height of approx. 2 mm above the bark, on zones covered by algal crust (sometimes with some Lecanora expallens). This made the measurements compatible. Measurements of temperature at even smaller distances would have been technically possible but would have caused too much time delay between the investigation of a trunk's climatic zones. The considered heights of 2 mm above the substrate are known to be well within the boundary layer even on wind-exposed, small and smooth bodies (WILLMER 1986 for upper sides of plant leaves).
Five locations per trunk face were investigated for temperature, four for wind and one for air-humidity measurements. At each location, measurements of the various microrelief zones were taken at directly neighbouring sites. Additionally, climate was also measured at a point that reflected the ambient macroclimate hardly modified by directly neighbouring substrates: at about 2 m distance from the respective trunk and also from other trunks, shrubs or buildings. Only in August and (for humidities) also September was the macroclimate not investigated.
The macroclimatic wind speed was also estimated on a Beaufort-scale (HEYER 1972). In further analyses of interactions with thermic and hygric conditions and with the animals' distribution, the Beaufort scale was a much more reliable parameter than the anemometer-measurements, which strongly fluctuated within minutes.
Animals were searched optically: first with a special lens from a distance of approx. 60 cm (7x magnification, 3.1 cm2 field of view), then with a hand-lens (10x magnification, 1.8 cm2 field of view). In each zone of exposure each microhabitat type present was sampled separately. Each sample consisted of ten randomly distributed direct counts covering 31 and 18 cm2 of microhabitat surface, respectively. For each single field of view the trunk was approached with careful movements.
Inner surfaces of microhabitats were examined exclusively with the hand lens and according to the following procedure: (a) The dark cavities of horizontal crevices and of Evernia prunastri thalli were illuminated with a small pen-light torch. At night, illumination was necessary for all types of microhabitats and was restricted to a single field of view and to less than about two seconds at a time. (b) Horizontal crevices were searched in a length that, multiplied by the approximate depth of the crevices, equalled approx. 18 cm2 (this length was measured in diameters of fields-of-view of the hand lens). Horizontal crevices were sometimes difficult to survey by eye. They were then additionally examined by gentle scraping with a large needle. (c) Thalli of the shrub-like lichen, Evernia prunastri, were opened in several layers with a large needle. (d) Vertical crevices were broken up with a knife and detritus was scraped off with a needle. (e) Investigation plots of crust-like lichens were completely scraped off on at least one in 10 trunks until mid-September. Later, when the encountered crusts became thicker, this was done for all crustose lichen plots.
For calculation of densities on the bark-surface-microhabitat types, the distance lens searches were only used in the bugs Temnostethus gracilis and Loricula elegantula and in the empidid fly Tachypezia nubila. For the remaining species, the hand lens searches were used. Animals found during distance lens searches were only exceptionally considered in addition - namely when no animals of this species were found during the subsequent hand lens counts of the respective microhabitat plot. This was to compensate for a possible chasing away of animals from the bark surface microhabitats into cavities caused by hand lens searching. The described situation occurred in less than 1 % of the investigated plots. This confirms more qualitative statements about the efficiency of optical searching for microarthropods on bark surfaces by MAYER (1957), BAUER (1979) and NICOLAI (1985). Hand lens counts even tended to be more efficient than distance lens counts, except in the mentioned bug and fly species and in Porcellio scaber (Isopoda). Many animals that had been spotted from a distance were tested by approaching them directly with the hand-lens, which did not induce any flight behaviour - except in the mentioned bug and fly species.
Most individuals of the Collembola, Psocoptera, Isopoda and Heteroptera could be identified in the field, most other animals had to be collected (minute exhaustor or wet brush) and were determined in the laboratory.
Approximate individual lengths were estimated in the field for most individuals of the Collembola Orchesella cincta (ca. < 1.7/ < 3/ > 3 mm), Entomobrya albocincta and Entomobrya nivalis (ca. < 0.8/ < 1.7/ > 1.7 mm), and the Psocopter Cerobasis guestifalica (ca. < 0.6/ < 1.2 / < 1.7 / > 1.7 mm). These individuals were termed "hatchlings" (smallest Cerobasis guestifalica), "juveniles", "middle-aged" and "adults", respectively. This classification was based on knowledge of the complete range of body sizes from laboratory-rearing. Proportions of "adult" Collembola were large in general, which corresponds to the long adulthood (i. e. maturity, decreased growth) of these animals (JOOSSE et. al. 1973). Animals found by distance lens searching were only considered for calculation of age class compositions when they were obviously different from those individuals found during the following hand lens searching (i. e. surplus animals, or such belonging to a different age class).
2.3. Sampling and processing of Evernia prunastri thalli
Large-scale photographs were taken (a) from August 1991 until April 1992 at intervals of 2 to 3 months in order to document the development of 27 thalli in 7 trunk areas (scale 1:1.25) and (b) in January and April 1993 (5 thalli in three trunk areas, scale 1.2:1). Simultaneously, thalli in an additional 11 ('91/'92) and 5 (‘93) trunk areas respectively were photographed using a smaller scale (1:2 to 1:4). Photographed thalli were growing on 5 oaks, one ash tree ('91/'92) and on three oaks ('93), respectively. Photos from 1991/1992 had previously been considered for qualitative observations in PRINZING (1992).
Five thalli, all strongly overgrown by Pleurococcus-algae and only sparsely grazed, located on solitary trees, were artificially injured in January 1993. Cortex- and phycobiot-layers were cut away with a scalpel along 2-4 rounded edges of branches in each thallus. The thalli were photographed immediately and again after a period of three months. For surgery and photographing (Leitz photo-macroscope M 400) three of the thalli were taken into the laboratory for one day.
The climatic exposures of thalli on the investigated +solitary tree trunks were determined according to the following rules: main wind directions at a trunk can be recognised from the shape of surrounding shrubs and tree-crowns ("flag trees", Holroyd 1970, Noguchi 1979). Tree exposure to a main wind-direction is due to shelter from surrounding hills, hedgerows and forest-edges (Barner 1983), which are very common in the areas of investigation. The wind-exposure below the crown increases with increasing height on the trunk (KERSHAW & LARSON 1974). Wind pulls off the substrate most strongly where the air-current can move along unhindered in a tangential direction (Häckel 1993, White, Mottershead & Harrison. 1992). This is the case on the two lateral trunk faces adjacent to the frontally wind-exposed face. At microscale, there is additional wind-shelter due to "valleys" of the bark-relief or to neighbouring thalli (Kershaw & Larson 1974, Nicolai 1985).
Solar radiation is independent of the height on the trunk, except in the areas shaded by the crown and at the very base of the trunk, which is more perpendicularly inclined to solar-radiation (Stoutjesdijk & Barkman 1992). On the other hand, there is a horizontal decline of solar-radiation from the S(-W)-exposed trunk-faces towards E- and W-faces (Geiger 1961, Nicolai 1985). The bark's microrelief does not steadily differentiate the exposure to sun because of its changing position throughout the day and the year (Nicolai 1985).
Precipitation is at its highest below those points where the crown drains snow and rain onto the trunk. Such drainage zones can be easily recognised from the strong cover by algae or even mosses on the bark (Matthey et al. 1989).
2.4. Processing of data and statistical analysis
2.4.1. Analysis of climate measurements
Temperature and wind speed means were calculated separately for each zone of exposure on each trunk. These means were the basis for all further calculations.
Water vapour pressure (e, in Pascal) was calculated from the measurements of temperature (T, in °C) and relative humidity (rh, in %) according to formulas given in HÄCKEL (1993):
![]()
![]()
Saturation deficit (SD, in Pascal) was calculated as:
![]()
The saturation deficit is the adequate expression for the atmospheric dryness that a poikilothermic small organism experiences (JOOSSE & GROEN 1970, BLOCK et al 1990, STOUTJESDIJK & BARKMAN 1992).
Wind distribution on the trunk surface was expressed as a decrease of windspeeds (m/sec) either (a) on the wind-sheltered as compared to the wind-exposed face of each trunk, comparing only equal wind exposures within the microrelief, or (b) on the sheltered as compared to the exposed microrelief zone within each trunk face.
Influences on the climate next to the trunk were analysed by multiple regression analyses (SYSTAT-computer-package, 1992). Such a procedure reveals the statistical influence of an "independent variable" on a certain "dependent variable", while keeping the effect of all the other independent variables included in the analysis constant. This effectively prevents pseudocorrelations within the data set (LAMPRECHT 1992). In 384 cases, measurements on all climatic variables were available, permitting a complete multiple regression analysis. The dependent variables were at least roughly normally distributed (test according to PAGE & PATTON 1991). This is more than necessary for a description of interactions by regression taking into account the several hundred samples investigated (WELKOWITZ et. al 1982, who suggest > 30 samples are sufficient). The independent variables are continuous as well as ordered categories (mostly binary). These can be successfully combined in multiple regression analysis (JONGMAN et. al. 1987).
The direct and indirect influences of an independent on a dependent variable were balanced by a path analysis: an indirect effect was considered as a "compound path". Its path coefficient is the product of the standardised partial regression coefficients of each of the involved direct interactions. The path coefficient of a multiple effect of an independent on a dependent variable is equal to the sum of the involved path coefficients (SOKAL & ROHLF 1994).
The univariate effect of categorial variables was tested by Kruskal-Wallis- and Mann-Whithney-U-tests (SYSTAT-computer-package, 1992).
In all cases p-values were two-tailed.
2.4.2. Analysis of the animals' use of different climates
When comparing the effect of climates on the arthropod species densities the effect of simultaneously changing microhabitat frequencies had to be taken into consideration and filtered out. The following scheme illustrates the applied solutions for this problem:
FIELD SITUATION (simple, extreme example, only 4 microhabitat types):
climate 1 climate 2
microhabitat mosaic in 3
sampled zones of expo-
sure per climate:
![]()
![]()
microhabitat types:
number of samples: 3 3 2 2 2 2 3 3
observed frequency of animals: 6 6 1 1 5 3 1 2
ß
ANALYSIS:
Graphical pots (used in Fig. 12 and 14): Average densities (and their standard errors) were calculated for each microhabitat type and summed up within each climate separately (equivalent procedures e. g. in BALDWIN & SCHMELZ 1996, GEBAUER et. al. 1995, OLSSEN & STAAF 1995):
5 indiv.
standardized climate standardized
microhabitat composition 1 2 microhabitat composition
Mean densities which were caused by only one to three animals of a rare species (< 70 individuals) found on a rare microhabitat type (Pertusaria albescens, vertical crevices, Evernia prunastri) were unreliably large compared to mean densities on other, more frequent microhabitat types. This could mostly be avoided by pooling the rare microhabitat type with another colonised microhabitat type that was more frequent and structurally similar to the respective rare microhabitat type (resulting categories of pooled microhabitats = "lichens", "crust cryptogams" or "crevices"). Otherwise, where this was not possible, the very few animals on the rare microhabitat type were omitted from the graph.
Statistical methods: 1) Expected frequencies of a species were calculated for each single microhabitat type under climates 1 and 2 adapted to the respective number of samples; e. g. in : 11 animals observed on 3 + 2 samples Þ frequencies expected under equal distribution = 6.6 + 4.4 (NIEMEYER 1974, MÜHLENBERG 1984). 2) These expected frequencies were summed within the climates 1 and 2 separately and the sums were tested against the sums of observed frequencies (Chi˛ test of goodness if applicable, otherwise Kolmogorow-Smirnow-test) (GLÜCK, pers. commun.):
|
climate |
||
|
1 |
2 |
|
|
S (observed frequencies.) |
14 |
11 |
|
S (standardised expected frequencies) |
14 |
11 |
Numbers of sampled animals effect the statistical significance of a pattern. This was compensated in the rare species (similar to suggestions by WIENS 1989): for sample sizes smaller than 80 (50) animals, Chi˛ values of > 2.9 (1.9) were still considered as relevant (equivalent to p < 0.09 (0.17), respectively, 1 df). With sample sizes < 30 animals, patterns of Chi˛ < 1.9 could not be interpreted as a valid rejection of H1-hypothesis and were not considered at all.
Samples were pooled within each category of the independent variable (FOWLER & COHEN 1986, MÜHLENBERG 1989).
Only diurnal samples were considered for comparisons between different weather conditions. Day-night-comparisons only included samples from months and areas of investigation where night excursions had been conducted (this equally applies to the following analysis of microhabitat use).
2.4.3. Analysis of the animals' microhabitat use
Tests were conducted on changes in relative densities (definition below) of animals per microhabitat type under two alternative climates (e. g. warmer vs. cooler trunk faces) by Chi2 tests of goodness, or by Kolmogorov-Smirnov test when expected frequencies were < 5 (LAMPRECHT 1992). Samples were pooled within each category of the independent variable (FOWLER & COHEN 1986, MÜHLENBERG 1989).
The relative density of a species on a given microhabitat "1", RD1, expresses the ratio between the absolute density on this microhabitat, D1, and the sum of absolute densities on all eight investigated microhabitats (indicated "1" - "8"). Thus, the relative density indicates the relative importance/suitability of "1" for the animals compared to the other microhabitats:
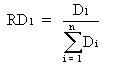 (n = 8) (1)
(n = 8) (1)
If the microhabitat "1" was of equal relative importance during two contrasting climates I and II, the expected relative densities (RDe) would be equal, too:
![]()
![]() (null hypothesis for the microhabitat "1")
(null hypothesis for the microhabitat "1")
or: 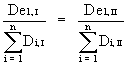 (De = expected density) (2)
(De = expected density) (2)
Since the density of animals on a microhabitat is calculated as the frequency F divided by the number of plots in which this microhabitat was found and investigated (sampling effort P), equation (2) can be rewritten as:
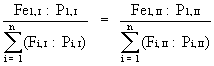 (Fe = expected frequency) (3)
(Fe = expected frequency) (3)
This equation allows the calculation of the null expected frequencies on "1" for the climates I and II, standardised for the differences in sampling efforts (caused by different microhabitat frequencies). Such standardised null expected frequencies were used for the subsequent Chi2 (Kolmogorov-Smirnov) test of goodness. Four steps were required (E. GLÜCK, H.-J. KRAMBECK, J. SCHIMMLER pers. comm.):
1) Rearrange equation (3) to:
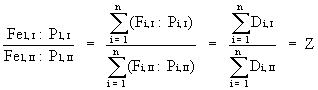 (4)
(4)
2) Replace the expected frequency for microhabitat "1" during one of the climates by the difference between the other expected frequency and the sum of the two observed frequencies on "1". This can be done, because the sum of expected frequencies must equal the sum of observed frequencies on a microhabitat:
![]() (
(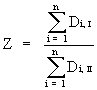 ) (5)
) (5)
3) Solve equation (5) for Fe1,I:
![]()
4) calculate the second expected frequency as:
![]()
Discriminant function analyses were performed (using a SYSTAT (1992) statistical package) in order to characterise the climates under which the different microhabitats are used by a species. Discriminant function analysis permits most straightforward interpretation of the results; it is robust against non-normality of data and optimal for the combination of binary and continuous variables (GILBERT 1968, MOORE 1973, GREEN 1979).
All statistical tests were two-tailed.
2.5. Determination and nomenclature were carried out according to GISIN 1960 (Collembola); GÜNTHER 1974 (Psocoptera); SELLNICK & FORSSLUND 1952 and SELLNICK 1960 (oribatid Carabodes labyrinthicus Michael); WAGNER 1961 (Anthocoridae); PÉRICART 1972 (Loriculidae); ROBERTS 1984 and HEIMER & NENTWIG 1991 (Araneae); GRUNER 1965/66 (isopods) and WIRTH 1980 (lichens). The oribatid Xenillus discrepans Grandjean was determined by G. WEIGMANN, the juvenile Carabodes by S. WOAS. The individuals called "Bdella cf semiscuta" Thor (trombidiformous Acari) largely resembled the descriptions by MICHOKA (1987), but might have to be redescribed by S. SWIFT.
2.6. Species list and sample sizes
The taxa that had been found on the investigated trees are listed up below. The rare species are often only mentioned as higher taxonomic units since the determinations were not yet tested by experts. Sample sizes are given in brackets for the more frequent species as the maximal number of animals that could be considered for further analysis of phenologies and distributions. The remaining species were each found in less than 25 specimens. The systematic scheme is not consistent for reasons of simplicity and partly due to a lack of consesus about the systematics. Nomenclature is in accordance with the references given in 2.5. and, for the other taxa, with BROHMER & TISCHLER (1982).
Oribatei (A cari): Carabodes labyrinthicus (161 animals); juvenile Carabodes (63)- most of them were probably juveniles of Carabodes labyrinthicus since other Carabodes species were only found in single individuals. Juvenile Carabodes are analysed separately from adult C. labyrinthicus, because they differ in shape, colour and stiffness of the cuticula, which are ecophysiologically relevant traits (KLIMA 1954,
1956).), Xenillus discrepans (54), Cymberamaeus cymba, Humerobates rostrolamellatus, Phauloppia lucorum, + ca. 19 further species
Trombidiformes (Acari): Bdella cf semiscuta (53), Bdellodes longirostris, Anystes vitis cf
Araneae: Linyphiidae: mainly Entelecara penicillata (83), Dictynidae, mainly Lathys humilis (29), Theridiidae; Clubionidae (largely juvenile); Segestriidae;
Philodromidae; Thomisidae;
Opiliones: Paroligolophus agrestis cf (many juveniles), Oligolophus tridens cf (ditto), three 3 further species
Myriapoda: Chilopda, Diplopoda including Polyxenus lagurus
Collembola: Orchesella cincta (375), Entomobrya nivalis (362), Entomobrya albocincta (503), Entomobrya corticalis, Anurophorus laricis cf (approx. 60, largely in winter, strongly clustered and mostly only in detritus of innermost bark crevices, not analysed), Isotomurus palustris cf (approx. 20, see Anurophorus laricis), Xynella spp., Lepidocyrtus cyaneus cf, Isotoma viridis, Folsomides spec., Symphypleona gen. spec.
Psocoptera: Cerobasis guestifalica (398), Reuterella helvimaculata (70), + eight further species
Dermaptera: Forficula auricula
Formicidae: mainly Lasius fuliginosus + three further species
Diptera - Brachycera: Dolichopodidae: Medetera jacula (test: H. MEYER) and two further, rare Medetera-species; Phoridae; Sciaridae; Scatophagidae; Xylophagidae; Rhagionidae, Empididae: Tachypezia nubila (34), Platypalpus spec.
Diptera - Nematocera: Psychodidae: possibly up to 15 species, Cecidomyidae, Bibionidae, Chironomidae
Thysanoptera: Limothriops caeralius cf. (> 40; largely in detritus of innermost crevices, many dead bodies, agricultural pest, using trunks for hibernation), two fruther species were seldomly found.
Hymenoptera: Braconidae; Ichneumonidae; Pachylommatidae; Proctotrupoidae; Cynipidae
Lepidoptera: Psychidae
Hemiptera: Aphididae, Psyllidae
Heteroptera: Anthocoridae: Temnostethus gracilis (30) + 3 further species,
Loriculidae: Loricula elegantula (37) + 2 further species
Miridae
Coleoptera: Lathridiidae; Curcolionidae; Staphyliniidae; Carabidae (mainly Dromius agilis)
Mollusca - Gastropoda: Succinidae; Clausilidae