http://www.collembola.org/taxa/collembo.htm
-
Last updated on
2024.10.31
by Frans Janssens
Peter F. Bellinger ( ),
Department of Biology, California State University, Northridge, CA 91330, USA
),
Department of Biology, California State University, Northridge, CA 91330, USA
Kenneth A. Christiansen ( ),
Department of Biology, Grinnell College, PO Box V3, Grinnell, IA 50112-0806, USA
),
Department of Biology, Grinnell College, PO Box V3, Grinnell, IA 50112-0806, USA
Frans Janssens,
Department of Biology, University of Antwerp, Antwerp, B-2020, Belgium
 |
|
Fig.1. Collembola habitus
(photographs 2000-2004 © Hopkin, S. (1);
2002 © Baquero, E. & Jordana, R. (2);
2004,2007 © Henderickx, H. (4);
2003 © Pettersson, B. (5);
2003 © Schoenherr, J. (6);
2004 © Baquero, E. (7);
2004 © Gielen, K. (8);
2004 © Vuijlsteke, M. (9);
2005 © Domene, X. (10);
2005 © Cheung, D. & Schmidt, J. (11);
2005 © Hall, K. (12);
2006-2007 © Stevens, M. (13);
2006 © Maddison, D.R. (14);
2008 © Ng, M. (15);
2008 © Baas, A.H. (16).
)
Citation suggested:
Bellinger, P.F., Christiansen, K.A. & Janssens, F. 1996-2024.
Checklist of the Collembola of the World. http://www.collembola.org
Introduction
Collembola are
small
([min. 0.12] 1-5 [max. 17] mm),
entognathous
(mouthparts, such as mandibulae and maxillae,
located within a 'gnathal pouch'),
wingless hexapods with antennae always present.
Most but not all Collembola may be recognised by a
posterior ventral forked abdominal appendage, the furca.
The presence of antennae
and absence of cerci distinguishes them from the other
entognathous hexapods, the
Protura
(with antennae and cerci absent) and the
Diplura
(with antennae and cerci or pincers present).
There are ca 9600 published species worldwide.
Collembolan fossils from the Devonian
(ca 400 million years ago)
are among the oldest known records of terrestrial animals.
These organisms
are virtually ubiquitous in terrestrial systems, ancient and thus,
one of the more successful arthropod lineages.
Etymology:
Lubbock (1870) proposed for the division of the Thysanura comprised
in the Linnaean genus Podura
the term Collembola, "as indicating the existence of a
projection or mammalia enabling the creature to attach or glue itself to the
body on which it stands" (Lubbock, 1873:36)
(from colla (Latin), from kolla (Greek): glue;
from embolon (Greek): that what has been thrown into something,
e.g. a wedge, a ram, a plug;
from emballein (Greek): to throw into, to insert).
This ventral projection, the ventral tube or collophore, plays an extremely
important role in the fluid and electrolyte balance.
The eversible vesicles of the ventral tube
may also be used as a source of 'grooming' fluid
and for adhering to smooth surfaces (after Hopkin, 1997:48-49).
In Anurida, that do not have a furca,
the eversible vesicles of the ventral tube may adhere to the surface waterfilm on which they can walk and deform it in such a way that
it is springloaded;
when the waterfilm is released the animal is launched upwards into the air
(Bush & Hu, 2006:351).
<!--<a href="../collembo.wav">kol-UM-bo-la</a>.-->
<!--<bgsound src="../collembo.wav" loop="1">-->
 |
Fig.2. Schematic diagram of the poduromorph body structure
(lateral view).
Modified after Potapov, M. in Babenko, A. (1988).
External anatomy and morphology:
The body of Collembola basically comprises three tagmata, a
head capsule, a
thorax with three segments, and an
abdomen with five segments and a terminal
periproct.
Thoracic and abdominal segments may be indistinct and may give the body a more
globular appearance.
The head bears two
antennae, two optional
postantennal organs, two optional
composed eyes and the
mouthparts.
The antennae principally consist of four articulations.
Antennal articulations may be subdivided or annulated.
Each composed eye consists of maximum eight ommatidia.
The mouthparts comprise the labrum, a pair of mandibulae, a pair of maxillae,
the hypopharynx and the bipartite labium.
The frontal labrum, the ventral labium and two lateral oral folds
enclose the other mouthparts in the buccal cavity (entognathy).
Each thoracic segment bears ventrally a pair of
walking limbs.
Each limb is made up of an
epicoxa,
subcoxa,
coxa,
trochanter,
femur,
tibia and
footcomplex,
the latter comprising the distal part of the tibia having a large outer lamellate unguis
and bearing a small inner unguicular tubercle with optional unguiculus or empodium
(Janssens, 1999-2010).
The anterior abdominal segment bears a
ventral tube or collophore having two eversible vesicles.
The third abdominal segment ventrally optionally bears the
retinaculum, a forked appendage that locks the springloaded furca
in place underneath the ventrum.
The fourth abdominal segment ventrally optionally bears the
furca.
The furca comprises the basal
manubrium, bearing two arms, each of them comprising a
dens and a
mucro.
The
genital orifice opens at the ventral side of the fifth abdominal segment.
The
anus opens terminally at the posterior abdominal
periproct.
The
linea ventralis
is a linear cuticular ventral groove that runs between the
base of the labium and the collophore (Hopkin, 1997:60).
Some discussion on morphological issues:
Tagmatisation in arthropodans is not monophyletic.
Assuming a primitive marine benthic crustacean discovering the potentials of
terrestrial habitats, tagmatisation is almost a natural process, since its main
effect is the localisation and specialisation of the locomotory system.
Improving the locomotory system is imperative for successfully invading
terrestrial habitats. On the other hand, cephalisation is just as
important. Both processes led to tripartite body division in arthropods
living in terrestrial habitats. In marine or freshwater habitats,
tripartite tagmatisation has no special advantadge.
 |
Fig.2a. Dicyrtomina saundersi
Left composed eye
2007 © Krebs, C.
 |
Fig.2a1. Dicyrtomina saundersi
Projected labial palps
2015.03.12 © Phillips, E.
The collembolan composed eye, with maximum 8 single eyes designated
as A to H (fig.2a), is derived from the compound eye of early crustaceans (Paulus, 1972).
Entognathy in Hexapoda s.l. is not
monophyletic. The collembolan entognathy might be developed
as an adaptation to terrestrial habitats.
Entognathy in crustaceans is rare. There are
some indications of an entognathic tendence in terrestrial
Amphipoda. This is interesting, because it might show that entognathy
is an evolutionary advantadge during the process of invading
terrestrial systems. Amphipoda are a more recent type of crustaceans:
oldest fossils are from the Eocene. So, they might be still in the
phase where early collembolan ancestors were in the preDevonian times.
The postantennal organs are the remnants of the 2nd pair of antennae
of its ancestral crustacean. The postantennal organs might be the specialised
sensory organ of the ancestral 2nd antennal apex that remained while the
2nd antenna shaft itself reduced (Lawrence, 1999).
To be completed.
Internal anatomy: introduction.
 |
Fig.2aa. Kiss of death. Entomobrya muscorum predated by Hybotidae. From Czechia. 2007.08.11 © Krásenský, P.
 |
Fig.2ab. Katiannidae. Spermatophores. From France. 2010.11.22 © Lebeaux, P.
Biology:
Development is direct with adults differing from juveniles in proportion,
size, pigment (usually juveniles are paler), and the absence of a genital
opening (Christiansen in Dindal, 1990:967).
In some genera a diapause occurs which may be associated with regressive
modification of mouthparts and digestive system and even striking external
modification of cuticle and the development of spines (ecomorphosis)
(Christiansen in Dindal, 1990:967-968).
Collembola moult throughout life with instars ranging from four to more
than 50 (Christiansen in Dindal, 1990:968).
Collembola are polyphagous, in general;
some species are saprophagous (decomposed plants), coprophagous (excrements),
necrophagous (cadavers), mycetophagous (fungi),
bacteriophagous (soil micro-organisms)
(Thibaud, 1970:103) or
pollinophagous (pollen).
Some are predacious.
In Sinella coeca and Sinella pouadensis, the adults eat their
own eggs, even when there is enough food supply
(Thibaud, 1970:132).
Collembola have separate sexes and indirect sperm transfer (Hopkin 1997:134).
Spermatophores (fig.2ab) are deposited
by the males on the substrate (Christiansen in Dindal, 1990:968), or
placed directly on the female genital opening (Hopkin 1997:134).
A variety of mechanisms have evolved to ensure successful 'capture' of this
spermatophore by the female (Christiansen in Dindal, 1990:968; Hopkin 1997:134).
 |
Fig.P. Ceratophysella sp. Predated by Aranea. From Belgium. 2020.02.16 © Huskens, M.-L.
 |
Fig.P2. Ptenothrix sp. Predated by Bdellidae. From the USA. 2020.12.28 © Pearson, R.R.
Predation.
Predators are represented by species of
Turbellaria
such as
Phagocata,
Chilopoda,
Opilionidae, Japygidae,
Arachnida
such as
Acari
(Erythraeoidea from Lithuania,
Bdellidae from the UK,
from the USA,
undefined mite from the UK),
Aranea
(e.g.
Salticidae
such as
Hentzia palmarum
or
Gen. spec. from the UK
or
Gen. spec. from the USA,
or
Ballus chalybeius from the UK,
or
Naphrys pulex from the USA,
or
Linyphiidae from the UK
or
Paidiscura pallens from the UK),
and
Gen. spec. from Belgium),
Pseudoscorpiones
(after Thibaud, 1970:105)
(such as
Neobisium muscorum from Belgium);
furthermore by Insecta such as
Hemiptera,
Coleoptera larvae,
Coleoptera: Pselaphinae,
Coleoptera: Staphylinidae,
Coleoptera: Rhyzobius litura,
Dolichopodidae (
from the UK,
from Sweden,
from the USA:South Carolina,
from the USA:Alaska),
Hybotidae,
and
Formicidae (
from the UK,
from France).
Physiology: introduction.
 |
Fig.3b. Podura aquatica and Sminthurides aquaticus, typically found on the surface of stagnant freshwaters. 2007.04.01 © Cornwall, N.J.
Ecology: Collembola
are soil and litter dwelling, preferring wet or damp surroundings.
Collembola inhabit soil and leaf litter, although
some species move actively over the surfaces of bark and flowers in
daylight.
They may be found in moss, under stones, in caves, in
ant nests
and
termite nests
but also in the intertidal zone on the coast, on
the surfaces of lakes and ponds or snow fields of glaciers.
Collembola are major
components of terrestrial ecosystems (and particularly significant
members of the soil communities), constituting a significant
proportion of the animal biomass and are thus frequently and easily
found. In forest soils they can reach densities of 200 to 1800 individuals per
dm3, densities only surpassed by the acarian
soil population (Handschin, 1955).
Abiotic factors:
In Hypogastruridae, the development is impacted as follows:
1. the lethal temperatures are -4°C and 28°C,
2. the optimum temperature range is 9°C to 12°C,
3. the hygrometric optimum is 98-100% relative humidity;
4. the lethal hygrometric minimun is 93% relative humidity
(Thibaud, 1970:161-173).
Paleontology: introduction.
Anthropological perspective:
Collembola can be pests principally by virtue of their presence
in the home.
But in many cases, the Collembola are just annoying 'guests',
a nuisance, rather than infestations causing a disease.
The infestations are classified as
domestic infestations (Collembola found in houses),
incidental human infestations (infestations through pot plants
in the bedroom, infestations by malfunctioning
pooter),
human infestations not associated with dermatitis and
human infestations associated with dermatitis.
In addition, one can also consider the
delusional infestations (psychotic infestations) and the
infestations due to 'sample contamination' (clinical errors, laboratory errors).
Phylogeny:
Handlirsch (1908) considers Collembola as a more or less
recent group of insects with an extreme specialisation. He considers
them as forms with a retrograde development reaching maturity while in
a larval state. (cited from Handschin, 1955:41,45).
Based on the discovery of the ca 400 million years old Devonian
fossil Rhyniella praecursor, and the
striking resemblance it shows with extant collembolan species, Tillyard (1928)
concludes that Collembola are primary, ancestral, and archaic
terrestrial arthropodans (cited from Handschin, 1955:41,49).
Gullan & Cranston (1994:192-194) consider Collembola
as the sistergroup of Insecta + Diplura,
grouped with Protura into Hexapoda.
Janssens & Lawrence (2002-2012)
propose that Collembola are highly specialised terrestrial Crustacea,
that have reached their evolutionary climax already in the Devonian,
when they dominated most terrestrial habitats.
The terrestrial competition between Collembola and early Insecta
might have triggered the latter to develop wings
to become 'masters in the sky' in the Carboniferous.
A phylogeny, applying the principle of total evidence,
using molecular and morphological characters,
strongly supports the monophyly of
Pancrustacea
(= Crustacea & Hexapoda)
(Giribet, Edgecombe & Wheeler, 2001:160).
Molecular phylogeny of the arthropods provide support
for a monophyletic Hexapoda/Branchiopoda clade
(Regier & Shultz, 1997:902,911).
Based on mitochondrial data, Lavrov et al. (2004) recover an (Insecta,
(Branchiopoda, Malacostraca)) clade and a (Collembola,
Maxillopoda) clade, which is confirmed by Cook et al. (2005)
(Cook, Yue & Akam, 2005:1301).
Physiological data show that Collembola evolved directly from marine
ancestors: haemolymph with high osmotic pressures and mainly composed of
inorganic salts (Little, 1983, 1990 cited from D'Haese, 2003:583).
So early crustaceans must have been adapted from marine habitats
in the Cambrium to terrestrial soil habitats in the Devonian.
Possibly, Collembola are derived from a benthic marine
maxillopod that explored the potentials of terrestrial soil habitats.
Methods: introduction.
Systematics:
The taxonomic hierarchy is mainly based on Bretfeld (1994, 1999),
D'Haese (2002:1148), and Deharveng (2004:427).
The systematics of the higher taxa that is presented here
is in line with some of the more 'recent' opinions.
Collembola are not considered as being Insecta
but as a taxonomic group with the same rank (class).
Note that also Protura and Diplura are currently classified as separate classes.
In an attempt to organise a combination in kind of harmony between
two by definition incompatible classification schools -
the Linnean school that uses a static, hierarchical system
with emphasis on the ranking of taxa
and the cladistic school that uses a dynamic, evolutionary system
with emphasis on the relationship between the taxa
-
the classification used here tries to map the more recent cladistic system
onto the conventional Linnean classification and ranking system.
Note that it will never be possible to combine both systems in
a 100% compatible way. In other words: different opinions and thus
classifications will continue to popup in the papers...
Hexapoda Blainville, 1816.
The finding of the reciprocal paraphyly of
Hexapoda and Crustacea suggests an
evolutionary scenario in which the acquisition of the hexapod condition may
have occurred several times independently in lineages descending from different
crustacean-like ancestors, possibly as a consequence of the process of
terrestrialisation
(Carapelli, Liò, Nardi, van der Wath & Frati, 2007).
Although found paraphyletic based on recent molecular studies, Hexapoda is
conveniantly maintained in the current taxonomic hierarchy
untill the disagreements between molecular and morphological analyses have
been resolved.
Apterygota Lang, 1889
(= Archaeognatha, Zygentoma, Diplura, Collembola and Protura)
is considered as being
an artificial assemblage of paraphyletic taxa
(Moen & Ellis, 1984)
and therefore not accepted anymore as a valid formal taxon by the
cladistic school of systematists (Hopkin, 1997:19)
(Bach de Roca, Gaju-Ricart & Compte-Sart, 1999:393).
Ellipura Börner, 1910 (= Collembola and Protura) is not a
monophyletic group (Bach de Roca, Gaju-Ricart & Compte-Sart, 1999:393) and
therefore not accepted in this classification.
- Superregnum Eucarya Woese, Kandler & Wheelis, 1990
- Regnum Animalia Linnæus, 1758
- Subregnum Eumetazoa Butschli, 1910
- Superphylum Ecdysozoa Aguinaldo AMA, Turbeville JM, Lindford LS, Rivera MC, Garey JR, Raff RA & Lake JA, 1997
- Phylum Arthropoda Latreille, 1829
- Subphylum Pancrustacea Zrzavy & Stys, 1997
- Superclassis Hexapoda Blainville, 1816
- Classis Collembola Lubbock J, 1870:295 sensu Bellini BC & al, 2022:14














 |
Ceratophysella meets Dicyrtomina
from the UK
2008.01.05 © Valentine., B
 |
Isotomurus palustris and Orchesella villosa
from the UK
2008.01.12 © Tonsbeek, M.
 |
Aggregation of Ceratophysella,
Hypogastrura and Proisotoma from the USA
2008.03.09 © Boeddeker, M.
- Ordo Neelipleona Massoud Z, 1971:198
- Familia Neelidae Folsom JW, 1896:391


 |
Neelus? sp.
2005 © Cheung, D. & Schmidt, J.
 |
Megalothorax? sp. nov. from the UK
2006 © Brocklehurst, K.
 |
Neelidae from the UK
2008.04.13 © Kilford., B
 |
Neelidae from Hungary
2010.03.09 © Pfliegler, W.
- Ordo Symphypleona Börner C, 1901:5 sensu Massoud, 1971


 |
Symphypleona from Holland
Sminthurinus aureus & Dicyrtoma fusca
2013.03.03 © van Duinen, J.
 |
Dicyrtominae juv. from the UK
Dorsally positioned gut; note dark gut contents
2014.08.15 © Phillips, E.
 |
Symphypleona from Spain
Escape jump
Disappearance act
2020.12.31 © Zeeders, M.
- Subordo Sminthuridida Bretfeld, 1986 sensu Sánchez-García A & Engel MS, 2016:4
- Superfamilia Sminthuridoidea sensu Fjellberg A, 1989:133
- Familia Sminthurididae Börner, 1906:163 sensu Schneider C & DHaese CA, 2023:795

 |
Mackenziella psocoides ♀ from Scandinavia
After Fjellberg, A, 2006 Fig.2
 |
Sminthurides aquaticus from Belgium
2000 © Hopkin, S.P.
 |
Sphaeridia serrata from the USA
2006 © Maddison, D.R.
- Subordo Appendiciphora Bretfeld, 1986 sensu Sánchez-García A & Engel MS, 2016:4
- Infrasubordo Katianniformia Bretfeld, 1986 sensu Sánchez-García A & Engel MS, 2016:4
- Superfamilia Katiannoidea Bretfeld, 1994
- Familia Katiannidae Börner, 1913:321 sensu Bretfeld G, 1999:13

 |
Two new Katiannidae from the UK
2009.06.06 © Ardron, P.A.
 |
Katiannina macgillivrayi from the USA
2009.11.26 © Justis, S.
- Familia Spinothecidae Delamare Deboutteville, 1961, sensu Bretfeld, 1994

 |
Adelphoderia regina from Tasmania
2014.03.10 © Murray, A.
 |
Adelphoderia regina from Tasmania
With neck organs
2014.03.10 © Murray, A.
- Familia Arrhopalitidae Stach, 1956, sensu Bretfeld G, 1999:13

 |
Arrhopalites hirtus from the USA
2005 © Cheung, D. & Schmidt, J.
 |
Arrhopalitidae from the USA
2024.04.05 © Hays, T.
- Familia Collophoridae Bretfeld G, 1999:13
 |
Collophora quadrioculata from Canada
2020.11.21 © Rennert, M.
 |
Collophora quadrioculata from Canada
Eyepatch reddish
4 ocelli
2020.11.21 © Rennert, M.
- Superfamilia Sturmioidea Bretfeld, 1994
- Familia Sturmiidae Bretfeld, 1994
 |
Sturmius sp. nov. from Panama
2009.03.16 © Palacios-Vargas, J.G.
 |
Sturmius sp. nov. from Panama
2009.03.16 © Palacios-Vargas, J.G.
- Infrasubordo Sminthuriformia Bretfeld, 1986 sensu Sánchez-García A & Engel MS, 2016:4
- Superfamilia Dicyrtomoidea Bretfeld, 1994
- Familia Dicyrtomidae Börner C, 1906:163, sensu Deharveng, L, 2004:427

 |
Dicyrtomina minuta f. ornata from the USA
Abdominal tubular wax excretions
2008.11.15 © Justis, S.
 |
Dicyrtomina ornata from Belgium
Eversed telescopic collophore vesicle
2017.02.03 © Huskens, M.-L.
 |
Dicyrtomidae from Mexico
After Murray A in Palacios-Vargas JG & Cruz-Leal JI, 2020 Fig.3
- Subfamilia Ptenothricinae Richards WR, 1968:25 sensu Bretfeld, G, 1999:128
 |
Ptenothrix sp. from Canada
Anterior dorsal spinelike setae
2008.12.11 © McCann, S.
 |
Ptenothrix sp. from Canada
Small abdomen with 3M+2N setae
M-setae in transverse row
2023.02.18 © Barbin, T.
 |
Ptenothrix sp. from Canada
Two unpaired midfacial setae
2023.12.17 © Chans, J.
- Subfamilia Dicyrtominae Richards WR, 1968:26 sensu Bretfeld, G, 1999:128

 |
Dicyrtomina sp. from New Zealand
2014.02.25 © Murray, A.
 |
Dicyrtoma fusca from Belgium
Abd.6 dorsally curved (*)
2016.02.19 © Huskens, M.-L.
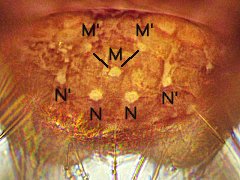 |
Dicyrtominae from Taiwan
Abd.6 with 3 M-setae + 4 N setae
M-setae form a distinct angle
2023.02.07 © Cheng, H.-J.
 |
Dicyrtomina ornata from France
More than 2 thick unpaired midfacial setae
2023.03.12 © Clerc, O.
- Superfamilia Sminthuroidea Bretfeld, 1994
- Familia Bourletiellidae Börner, 1913:322, sensu Bretfeld, 1994
 |
Deuterosminthurus pallipes ♂ & var. bellingeri ♀ from France
Courtship ritual
2010.06.04 © Elpachato.
 |
Bourletiella sp. from Australia
2012.07.03 © Hort, J.
 |
Bourletiella hortensis ♀ from Belgium
5 bothriotricha ABC+DE
2020.07.30 © Huskens, M.-L.
- Familia Sminthuridae Lubbock, 1862, sensu Deharveng, L, 2004:427


 |
Sminthuridae from Chile
2022.03.19 © Gargiulo, M.
- Subfamilia Sphyrothecinae Betsch J-M, 1980:149
 |
Neosminthurus richardsi from the USA
2008.01.26 © Gross, J.
 |
Neosminthurus sp. from the USA
2019.06.22 © Crockwell, J.M.
- Subfamilia Sminthurinae Lubbock, 1862, sensu Deharveng, L, 2004:427


 |
Sminthurinae from Russia
2006 © Macroclub.ru.
 |
Sminthurus sp. nov. from the USA
2007.04.30 © Cowen, R.
- Subfamilia Songhaicinae Sánchez-García A & Engel MS, 2016:12 sensu Bernard EC & Wynne JJ, 2017:77

- Ordo Poduromorpha Börner C, 1913:318, sensu D'Haese CA, 2002:1148



- Superfamilia Onychiuroidea sensu D'Haese CA, 2002-2003

- Familia Onychiuridae Lubbock J in Börner C, 1913:319


- Subfamilia Tetrodontophorinae Stach, 1954

 |
Tetrodontophora bielanensis from Germany
2004 © Hopkin, S.P.
 |
Homaloproctus sauteri from Japan
Eyes absent
2014.01.05 © Hara, Y.
- Subfamilia Lophognathellinae Stach J, 1954:6

 |
Lophognathella choreutes from Canada
2015.08.09 © Murray, A.
 |
Lophognathella choreutes subadult from the USA
2017.11.29 © Dobbs, Br.
- Subfamilia Onychiurinae Börner C, 1901:698


 |
Onychiurinae from China
Pigment and eyes absent
2008.07.17 © NCode, A.
 |
Onychiurinae from Greece
Ventral furca absent
2012.11.14 © Henderickx, H.
 |
Onychiurinae from France
Cave species
2014.05.07 © Alonso, C.
 |
Onychiurinae from Croatia
Ant.3 sensorial organ (*) well developed
2019.03.dd © Lukic, M.
 |
Onychiurinae from Belgium
Anal spines
2020.09.27 © Huskens, M.-L.
 |
Onychiurinae from Belgium
Post-antennal organ (PAO) in groove
2020.12.16 © Huskens, M.-L.
 |
Onychiurinae from Taiwan
Egg cluster
2021.12.09 © Cheng, H.-J.
- Familia Tullbergiidae Bagnall RS, 1935:238




 |
Tullbergiidae from the USA
Body narrow worm-shaped
2012.02.16 © Kouri, J.
 |
Tullbergiidae from the UK
Pigmentation absent, eyes absent
2012.10.02 © Murray, A.
 |
Tullbergiidae from Holland
Antennae short, legs short
2020.09.28 © Kamsteeg, G.
- Familia Isotogastruridae Thibaud J-M & Najt J, 1992:545 i.s.
 |
Isotogastruridae from South Africa
2011 © Deharveng, L.
- Familia Pachytullbergiidae Stach, 1954:6
- Familia Odontellidae Massoud Z, 1967:61

 |
Odontella sp. from the USA
2006 © Bernard, E.
 |
Superodontella cornifer from the USA
Distinctly conical antennae
2016.11.02 © Dobbs, Br.
 |
Superodontella lamellifera juv. from France
With conical antennae
2019.06.10 © Garcelon, P.
- Superfamilia Hypogastruroidea Salmon JT, 1964:103, sensu Deharveng L, 2004:427

- Familia Hypogastruridae Börner, 1906

 |
Hypogastrura sp. from the USA
Mass aggregation
2008.02.12 © Boeddeker, M.
 |
Hypogastruridae from Peru
2009.01-03.dd © Damiano Palomino., B
 |
Hypogastrura sp. from the USA
With uplifted abdomen and extended furcula
2009.02.26 © Cowen, R.
 |
Hypogastruridae from the USA
Short furcula with broad manubrium
2012.03.21 © Kouri, J.
- Familia Paleotullbergiidae Stach, 1954:6 sensu Deharveng L, 2004:427
- Superfamilia Poduroidea sensu Palacios-Vargas, 1994:409
- Familia Poduridae Latreille, 1804, i.s.
 |
Podura aquatica from Belgium
2001 © Hopkin, S.P.
 |
Podura aquatica from Belgium
2006.03.26 © De Wilde, A.
- Superfamilia Neanuroidea Massoud Z, 1967:58, sensu D'Haese CA, 2002:1148

- Familia Neanuridae Börner, 1901 sensu Yosii, 1956

 |
Neanuridae from New Zealand
2006 Minor, M., & Robertson, A. © SoilBugs
 |
Neanuridae from the USA
2009.06.22 © Light, K.
- Subfamilia Caputanurininae Lee B-H, 1983:28

 |
Caputanurina serrata from Korea
SEM dorsal
After Lee B-H, 1983 Fig.3A
- Subfamilia Frieseinae Massoud, 1967
 |
Friesea sp. from Belgium
2010.01.05 © Soors, J.
- Subfamilia Pseudachorutinae Börner C, 1906:160
 |
Pseudachorutinae from Australia
2017.03.04 © Steve & Alison.
 |
Pseudachorutinae from the USA
2017.10.16 © Johnson, S.T.
 |
Pseudachorutinae from Singapore
2021.11.29 © Bay, N.
 |
Pseudachorutinae from Taiwan
Pointed mouthcone
2022.04.09 © Cheng, H.-J.
- Subfamilia Uchidanurinae Salmon JT, 1964:109 sensu Greenslade P, 2015:5
 |
Denisimeria longilobata from Malaysia
After Massoud Z, 1967 Fig.74
- Subfamilia Morulininae Yosii, 1961 sensu Cassagnau, 1983
 |
Morulina multatuberculata from the USA
With large morula-like postantennal organ (PAO)
2009.03.03 © Roffler, D.
 |
Morulina sp. from the USA
2013.07.05 © Brown, M.H.
 |
?Morulina? sp. from China
2023.03.03 © El-Rady, J.
 |
Morulina sp. from the USA
With large morula-like postantennal organ
Notice curled-up phoretic Nematoda
2024.03.25 © Coogler, J.
- Subfamilia Neanurinae Börner C, 1901:33 sensu Cassagnau, 1989



 |
Neanurinae from Singapore
2007 © Anker, A.
 |
Neanurinae from Croatia
2008.05.25 © Keresztes, G.
 |
Neanurinae from China
2012.10.18 © Deart, Y.
 |
Neanurinae from Singapore
2020.06.15 © Luna, J.
- Familia Brachystomellidae Stach, 1949


 |
Brachystomella parvula from the UK
2012.06.11 © Murray, A.
 |
Brachystomellidae from Tasmania
2014.04.11 © Murray, A.
- Superfamilia Gulgastruroidea
- Familia Gulgastruridae Lee B-H & Thibaud J-M, 1998:453
 |
Gulgastrura reticulosa from Korea
SEM
After Lee B-H & Thibaud J-M, 1987 Fig.1
- Ordo Entomobryomorpha Börner, 1913:319, sensu Soto-Adames FN et al., 2008:501



- Superfamilia Tomoceroidea Szeptycki A, 1979:112

 |
Tomocerus minor from the UK
Large rounded body scales
2009.09.22 © Robertson, A.
- Familia Oncopoduridae Carl J & Lebedinsky J, 1905:565

 |
Oncopodura sp. nov. from Belgium
2005 © Janssens, F.
 |
Oncopodura crassicornis from the UK
Abd.4 subequal to abd.3
2013.11.30 © Murray, A.
- Familia Tomoceridae Schäffer, 1896


 |
Pogonognathellus sp. from the UK
Body covered with iridescent scales
2007.12.19 © Campbell, A.
 |
Tomocerus minor from the UK
3rd abdominal segment superequal to 4th
2008.09.24 © Robertson, A.
- Superfamilia Isotomoidea Szeptycki, 1979:112 sensu Soto-Adames FN & al, 2008:504

- Familia Actaletidae Börner, 1902, sensu Soto-Adames FN et al., 2008:506

 |
Actaletidae from Mexico
2002 © Palacios-Vargas, J.G.
- Familia Isotomidae Schäffer, 1896


 |
Isotomidae from Portugal
2012.10.16 © Murray, A.
- Subfamilia Proisotominae Stach, 1947:6
 |
Proisotominae from Tasmania
2014.04.08 © Murray, A.
 |
Proisotominae from Tasmania
2014.04.16 © Murray, A.
- Subfamilia Anurophorinae Börner C, 1901:42




 |
Anurophorus sp. from the USA
2009.11.21 © Thombs, D.
- Subfamilia Isotominae Schäffer, 1896
 |
Isotominae from Belgium
2006 © Vuijlsteke, M.
 |
Isotoma viridis from the USA
2009.03.10 © Cowen, R.
 |
Isotominae gen. nov. from Croatia
Close to Isotomurus
2019.03.dd © Bedek, J.
- Subfamilia Pachyotominae Potapov MB, 2001:18
- Familia Protentomobryidae Folsom, 1937, -
- Superfamilia Entomobryoidea Womersley, 1934 sensu Godeiro NN et al, 2022:106


- Familia Orchesellidae Börner C, 1906:162 sensu Zhang F et al, 2019:273




 |
Orchesella sp. from Estonia
2006.10.06 © Tartes, U.
 |
Heteromurinae from Australia
2009.09.25 © Daley, A.
 |
Orchesellidae from Chile
2021.08.29 © Gargiulo, M.
- Subfamilia
Nothobryinae Zhang F & Deharveng L, 2015:307 sensu Nunes & al, 2020:27 



- Subfamilia Bessoniellinae Soto-Adames FN & al, 2008 sensu Zhang F & Deharveng L, 2015:307
- Subfamilia Heteromurinae Absolon & Kseneman, 1942 sensu Zhang F et Deharveng L, 2015:307



 |
Heteromurinae from Australia
2009.09.25 © Daley, A.
- Subfamilia Orchesellinae Börner C, 1906:162 sensu Zhang F & Deharveng L, 2015:307


 |
Orchesella sp. from Estonia
2006.09.17 © Tartes, U.
 |
Orchesella cincta from the UK
With subdivided 2 basal antennomeres
2008.09.21 © Robertson, A.
Familia Praentomobryidae Christiansen, KA et Nascimbene, P, 2006:354,- 
Familia Entomobryidae Womersley, 1934 sensu Godeiro NN et al, 2022:106 partim 


- Subfamilia Willowsiinae Yoshii R & Suhardjono YR, 1989:35




 |
Willowsia buski from France
2007.06.09 © Lebeaux, P.
 |
Willowsia platani from the UK
2007.06.30 © Cornwall, N.J.
- Subfamilia Entomobryinae Schäffer, 1896 sensu Godeiro NN et al, 2022:106



 |
Entomobryinae from the USA
Entomobrya griseoolivata
E. assuta, E. atrocincta ♂
2006 © McClarin, J.
 |
Entomobrya intermedia from the UK
Two blunt thick mesothoracic sensilla
2015.09.11 © Murray, A.
- Subfamilia Salininae Absolon & Kseneman, 1942:24 sensu Godeiro NN et al, 2022:106


 |
Salininae from Taiwan
2010.06.23 © Chien, H.-C.
 |
Salininae from Australia
2012.06.23 © Kathy & Marissa.
Familia Paronellidae Börner, 1906:162 sensu Zhang F & al, 2019:275 partim sensu Janssens F, 2023 

 |
Paronellidae from China
2008.07.18 © NCode, A.
 |
Paronellidae from Taiwan
2009.02.04 © Wu, S.
- Subfamilia Paronellinae Börner C, 1906:162 sensu Godeiro NN et al, 2022:106



 |
Paronellinae from Taiwan
2x abd.1-3 <= abd.4
2023.10.22 © Cheng, H.-J.
- Subfamilia Troglopedetinae Börner C, 1913:321
- Subfamilia Seirinae Yosii R, 1961 sensu Zhang F & al, 2019:274


 |
Seirinae from Vietnam
2014.01.17 © Bertner, P.
 |
Seirinae from Malaysia
?With cercus?
2014.02.08 © Bertner, P.
- Subfamilia Lepidocyrtinae Wahlgren E, 1906:67 sensu Zang F et al, 2019:252

 |
Lepidocyrtinae from Sweden
2006.03.25 © Hall, K.
 |
Lepidocyrtinae from Singapore
2014.01.22 © Bay, N.
Familia Oncobryidae Christiansen, KA et Pike, E, 2002:167,-
Superfamilia Coenaletoidea Soto-Adames FN et al., 2008:506
- Familia Coenaletidae Bellinger PF, 1985:117
 |
Coenaletes vangoethemi ♂ from New Guinea
Abd.3-4 fused
After Bellinger PF, 1985 Fig.13
 |
Coenaletes caribaeus
2006 © Palacios-Vargas, J.G.
Familia ludens
Familia Fuzzballidae Janssens, 2006
- Genus Pareidolia Janssens, 2008
- Species ramosi Janssens, 2008
 |
Pareidolia ramosi from the USA
2006.02.06 © Ramos, K.
- Species bloombergi Janssens, 2013
 |
Pareidolia bloombergi from the USA
2013.04.15 © Bloomberg, M.
Fig.4. Tentative ordinal phylogenetic relationships
+----- Entomobryomorpha s.s.
+-A-+
| +----- Poduromorpha
+-eC-+
| | +----- Symphypleona s.s.
| | |
<--C--+ +-S-+ +-- Sminthuridoidea
| +--+
| +-- Coenaletidae
|
+-------------- Neelipleona
|
This ordinal tree (Fig.4) is based
on the views of relationships among
orders of Collembola based on phylogenies proposed by Börner (1901),
Cassagnau (1971),
Massoud (1971, 1976) Moen & Ellis (1984), Bretfeld (1986), Fjellberg (1994),
Soto-Adames (1996), D'Haese (2002, 2003), Park (2002), Deharveng (2004),
Gao & al. (2008), Xiong & al. (2008), Schneider & al. (2011),
Zhang & Deharveng (2015), Yu & al. (2016), Leo & al. (2019), Zhang & al. (2019),
Cucini & al. (2020), and Bellini & al. (2022)
(see also Bellini & al. (2023)).
Traditionally, the Collembola have been divided into 6 groups
(Arthropleona, Poduromorpha, Metaxypleona, Neelipleona, Entomobryomorpha,
and Symphypleona)
which different authors have considered to represent orders, sections or every
category in between these two.
D'Haese (2002) and Xiong & al. (2008)
considered Entomobryomorpha as paraphyletic
and D'Haese (2002:1148) proposed
Tomoceromorpha (= Tomoceroidea) as a new basic group of Collembola.
Schneider & al. (2011) and Leo & al. (2019)
confirmed 1. the monophyly of Neelipleona
and 2. that Neelipleona are not closely related to Symphypleona.
Zhang & Deharveng (2015), Leo & al. (2019, 2019)
and Nardi & al. (2020)
concluded that Entomobryidae resp. Entomobryomorpha
form a phylogenetic cluster with Symphypleona.
Yu & al. (2016), Leo & al. (2019) and Cucini & al. (2020)
consider Neelipleona and Poduromorpha closely related.
Zhang & al. (2019)
concluded that Poduromorpha is basic to Entomobryomorpha and Symphypleona,
and considered Tomoceridae as basic sistergroup of all other Entomobryomorpha.
Nardi & al. (2020)
found that Sminthurididae are basal to all other Symphypleona.
Based on Cucini & al. (2020),
Tomoceromorpha of D'Haese (2002) is polyphyletic :
Tomocerinae clusters with Symphypleona s.l. (S),
while the remaining
Tomoceromorpha (Lepidophorellinae + Oncopoduridae = Oncopoduridae s.l.)
forms a cluster with Neelipleona,
which is sister to Poduromorpha.
The phylogenetic hypothesis of Cucini & al. (2020) implies that
the reduction of the protergite occured independently 3 times.
Fig.5. Tentative ordinal evolutionary timeline
+-------- Entomobryomorpha
|
<--C--+----+--A--+-------- Poduromorpha
| |
| +-------------- Symphypleona
|
+------------------- Neelipleona
|
The mitogenomic phylogeny of Bellini & al. (2022)
retrieved the more traditional tree
(Neelipleona + (Symphypleona (S) + Arthropleona (A))(=Eucollembola (eC)))
with Neelipleona as most basal group.
The Arthropleona of Börner (1901) have been revived
although with poor node support.
We include Coenaletidae tentatively in Symphypleona given they share
fused abdominal segments and grasping antennae as common features.
We consider Coenaletidae (= derived Tomoceromorpha with grasping antennae)
as extant representatives of transient forms between
Tomocerinae and Symphypleona,
and as the sistergroup of the Symphypleona with grasping antennae :
Sminthuridoidea (= Mackenziellidae + Sminthurididae).
A tentative evolutionary timeline (Fig.5) has been deduced from
Bellini & al. (2022) and has been taken into account in the current
taxonomic hierarchy. The Poduromorpha are considered plesiomorph
given they have the least evolutionary changes (mutations) compared to the
common Collembola ancestor.
Acknowledgements
We would like to thank, in chronological order,
Louis Deharveng, Steve Hopkin, Toby Barton and Robert Vargovitsh for their
constructive comments.
Notes
Note on the authority of Onychiuridae.
In the papers, there is no uniform opinion on the authorship and year of
the family Onychiuridae.
Taxonomists give in their papers 'Lubbock, 1867', 'Lubbock, 1871',
'Börner, 1901', 'Börner, 1913', or simply avoid mentioning the author and year.
To solve this ambiguity it is required to go back to the source : Börner (1913).
Börner 1913:319 published 'Onychiuridae' with authority clause '(Lbk., CB.)'.
The meaning of this authority clause can be deduced from
'Isotomidae (Schffr., CB. 1896-1903)' (also on p.319).
Indeed Schäffer defined 'Isotominae' in 1896
and Börner raised it to family rank in 1903.
Therefore '(Schffr., CB. 1896-1903)' means 'Schäffer 1896 sensu Börner 1903'.
In analogy, '(Lbk., CB.)' then means 'Lubbock sensu Börner'.
So, 'Onychiuridae (Lbk., CB.)' = 'Onychiuridae Lbk. sensu CB'.
Given Börner published the family name in 1913:
'Onychiuridae Lubbock sensu Börner 1913'.
Börner considered 'Lipuridae Lubbock 1869(sic)' as synonym of 'Onychiuridae'.
Lipuridae of Lubbock was invalid given it was based on Lipura Burmeister 1838
which was homonomous with Lipura Illiger 1811.
So Börner attributed Lubbock post mortem as author of Onychiuridae,
that replaced the invalid Lipuridae of Lubbock.
To conclude :
Given the family name 'Onychiuridae' was
attributed to Lubbock by Börner in 1913,
in this work, we use the following authority clause
'Onychiuridae Lubbock in Börner 1913:319'.
This authority clause reflects and respects Börner's intention
to honour Lubbock as author of the family.
References
![]() ),
Department of Biology, California State University, Northridge, CA 91330, USA
),
Department of Biology, California State University, Northridge, CA 91330, USA
![]() ),
Department of Biology, Grinnell College, PO Box V3, Grinnell, IA 50112-0806, USA
),
Department of Biology, Grinnell College, PO Box V3, Grinnell, IA 50112-0806, USA


















































